 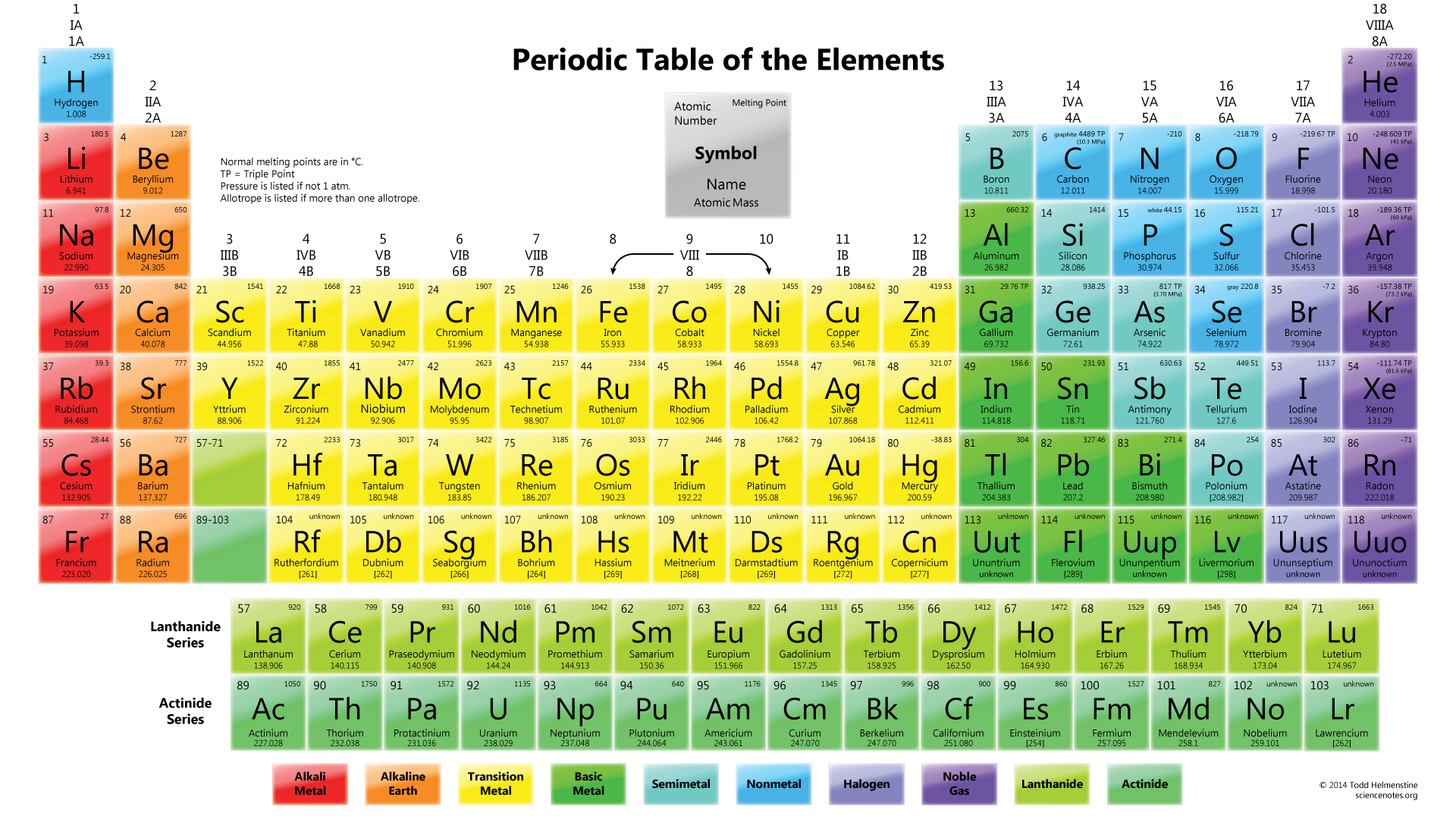
But, most importantly, the atomic number is the same as the number of protons in the nucleus of an atom of an element, as well as the same amount of electrons that surround the nucleus, when it is in a neutral state. The atomic number represents exactly where in the periodic table an element stands. When you move sequentially from the top left to the bottom right across the periodic table, the atomic number increases. Hydrogen has an atomic mass of 1.0079 and uranium has an atomic mass of 238.029. The number at the bottom represents the atomic mass. For example, the symbol for mercury is Hg. Generally, the atomic symbol correlates to the name, as C relates to carbon, but sometimes they do not. It is important to know the atomic symbol, because not all tables have the names listed below plus, that is how scientists notate the elements in their writing. So for hydrogen it is H and for uranium it is U. The letter in the middle is the atomic symbol. The atomic number for hydrogen is 1, and the atomic number for uranium is 92. The number at the top of the box represents the atomic number. Now, let’s look at all the stuff inside the individual boxes. The horizontal rows, the ones that run side to side, are called periods. The vertical columns, the ones that run up and down, are called groups. Now, let’s look at how to read the periodic table. Since, the table has seen small changes, and many, many elements have been added. In 1879 Mendeleev’s periodic table became widely accepted, when it predicted the existence of gallium, germanium, and scandium. The exciting discovery that physical and chemical characteristics of the elements are periodic functions of their atomic weight became known as the periodic law. Among heavier elements, he found that characteristics recurred every seventeen elements. He found that elements with low atomic weights had similar chemical characteristics that recurred every seven elements. By simply leaving openings for elements that he believed were undiscovered, he developed an organizational scheme that vertically grouped elements with alike properties. He realized that there was a number of unknown elements with atomic weights between elements that had already been discovered. Mendeleev noticed something that no one else had. In 1869, Russian chemist Dmitry Mendeleyev systematically arranged elements in a table according to their atomic weights. Because of this theory, Dalton created a scale of atomic weight based on the hydrogen atom. In 1803 English chemist, John Dalton, used the general scientific recognition, that elements combined with each other according to different ratios by weight, to create an atomic theory that claimed all elements were built out of variable numbers of hydrogen atoms. He then proceeded to compile a list of 33 elements, which just so happen to not actually be elements but he did spur this idea of systematizing and understanding elements. In 1789, the French chemist Antoine Laurent Lavoisier defined an element as a fundamental substance that could not be broken down by any chemical means then known. Let’s look at a short history of the Periodic Table. Through its innovative organizational layout, the table gives us concise and fundamental information not just about every single element, but also about trends that occur across all the elements. %20Periodic%20groups%20and%20periods.jpg)
The periodic table of elements is, hands down, one of the most important tools you will use in chemistry. Hey, guys! Welcome to this Mometrix video over the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
